Nitrogen Management
Nitrogen has a special role among minerals in the soil: plants need large quantities to achieve high quality and yield
Nitrogen is the fourth most common element in living tissue, after carbon, hydrogen and oxygen. It is an essential element in amino acids and a key component of chlorophyll, DNA and RNA. Without nitrogen there can be no life: no organ regeneration, no plant development or fruit and seed formation — and ultimately no yield. This is why nitrogen is commonly referred to as the engine of plant growth.
Nitrogen fertilisers can be applied in many different forms. These nitrogen-based fertilisers can be artificial (including urea and ammonium nitrate) or organic manures (including cattle slurry, pig slurry and digestate from anaerobic digesters).
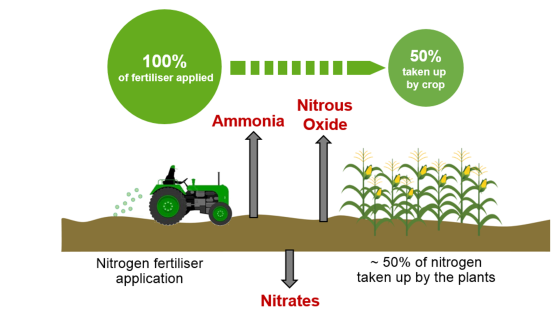
Only around 50% of applied nitrogen is taken up by crops
The rest is lost to the air and water or bound to the soil. Nitrogen losses can occur in the form of ammonia, nitrate leaching and the release of nitrous oxide into the atmosphere. While these losses generally result in an economic cost on farm, they also have a negative impact on the environment.
Effect of Nitrogen losses:
Ammonia losses
Significant ammonia losses can occur after the application of urea-based fertilisers. Ammonia is an air pollutant. A key component of smog, it binds with other pollutants and particles in the atmosphere, leading to negative impacts on human health. It is carried long distances by wind and brought down with rainfall, acting as a nitrogen fertiliser far from where it was intended and damaging sensitive natural habitats.
Nitrate leaching
Nitrate is highly mobile in the soil. After heavy rainfall or low plant uptake, nitrate can leach out of the soil profile and accumulate in groundwater, which can be toxic if threshold limits are exceeded. Nitrate in surface water bodies stimulates water plant and algae growth. As algae and/or water plants decay, the resulting oxygen depletion may, under extreme conditions, lead to mortality in fish populations.
Nitrous oxide losses
Nitrous oxide occurs during nitrification (conversion of ammonium into nitrite and nitrate through soil bacteria). Next to carbon dioxide and methane, nitrous oxide is one of the most dangerous greenhouse gases. Its global warming potential is 298 times that of carbon dioxide. Even small nitrous oxide losses may represent a cost factor to growers as well as a negative environmental impact.
Nitrogen stabilisers:
Nitrogen use efficiency technologies can help growers improve flexibility and efficiency with their fertiliser programmes so they can better protect their investments. There are two important technologies:
Urease inhibitors prevent the conversion of urea into ammonium by temporarily blocking extracellular enzymes in the soil called urease enzymes.
Nitrification inhibitors delay the further transformation of ammonium into nitrate.
These solutions can make a real impact. More nitrogen is available to crops over a longer window of time leading to benefits such as:
- Improved efficiency
- Greater certainty
- Improved environmental footprint


